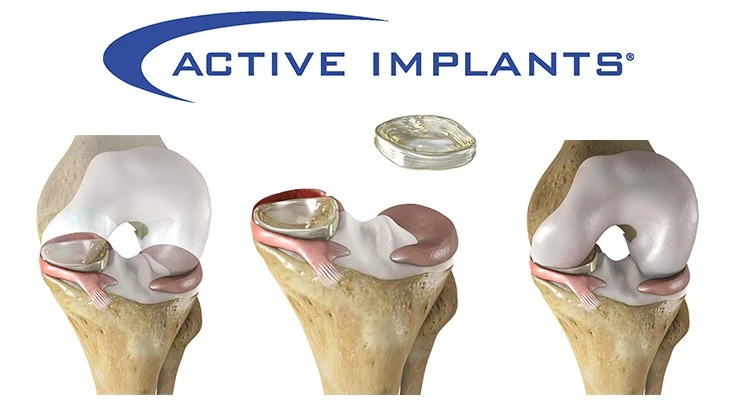
Active Implants’ NUsurface meniscus implant has been granted a Breakthrough Device Designation from the U.S. Food and Drug Administration (FDA). The NUsurface Meniscus Implant is the first artificial meniscus to be marketed in Europe and, if cleared by the FDA, would be the first artificial meniscus in the U.S.
The FDA Breakthrough Devices Program was implemented to expedite the development and review process for medical devices that are novel or offer new technology for patients with life-threatening or irreversibly debilitating conditions. This program is designed to ensure patients and healthcare providers have more timely access to vital devices.
“The Breakthrough Device Designation is a significant step in our mission to fulfill a substantial unmet need in the U.S. orthopedic market,” says Ted Davis, president and CEO of Active Implants. “We believe we will have the data required for our FDA submission next year, after completing enrollment in our clinical trials in 2018. We look forward to working closely with the FDA to expedite the review process for the NUsurface Implant to provide a new treatment option to the hundreds of thousands of patients who continue to experience persistent knee pain following meniscus surgery, but are too young for total knee replacement.”
Latest from Today's Medical Developments
- IMTS 2026 runs Sept. 14-19 at McCormick Place in Chicago, Illinois
- Master Bond’s MasterSil 800Med
- ZEISS celebrates 100 years of advancing innovation in the US
- Teleflex sells acute care and urology businesses for $2.03 billion
- HANNOVER MESSE: Where research and manufacturing meet
- What’s next for the design and manufacturing industry in 2026?
- Arcline to sell Medical Manufacturing Technologies to Perimeter Solutions
- Decline in German machine tool orders bottoming out





